Behavioral and Mental Health
Discover how Main Line Health helps those who struggle with mental illness and substance use live healthy and fulfilling lives.
Many people living with symptoms of major depression have tried multiple depression medications or combinations of antidepressants. Although the medications can be effective for some patients, they are systemic (circulate in the bloodstream). This often causes undesirable side effects such as weight gain, sexual dysfunction, upset stomach, sleepiness and dry mouth.
Through Main Line Health's comprehensive behavioral health program, we provide outpatient transcranial magnetic stimulation (TMS) therapy for depression at Mirmont Outpatient Center in Broomall.
In clinical trials, one in two patients experienced significant improvement in symptom and one in three reported a complete resolution of their symptoms. [1]
TMS was included in the 2010 American Psychiatric Association (APA) Practice Guideline for the Treatment of Patients with Major Depression. These guidelines provide recommendations for how psychiatrists treat depression. It is also an FDA-approved treatment and has proven effective for patients with severe depression whose condition has not improved from use of antidepressants. TMS works by using magnetic pulses precisely targeted to areas of the brain associated with mood, which are underactive in depressed people.
Transcranial magnetic stimulation therapy is a noninvasive procedure which does not require sedation. It works by positioning an electromagnetic coil over very precise areas of the scalp and sends targeted magnetic impulses, similar to an MRI, which stimulate regions of the brain associated with mood control and depression. The majority of patients that receive TMS treatments for major depressive disorder experience significant and long-lasting improvement compared to when they began treatment.[2]
After TMS treatments, your doctor will discuss a long-term care plan which usually includes a combination of medication and follow-up visits. Some patients may need additional treatments.
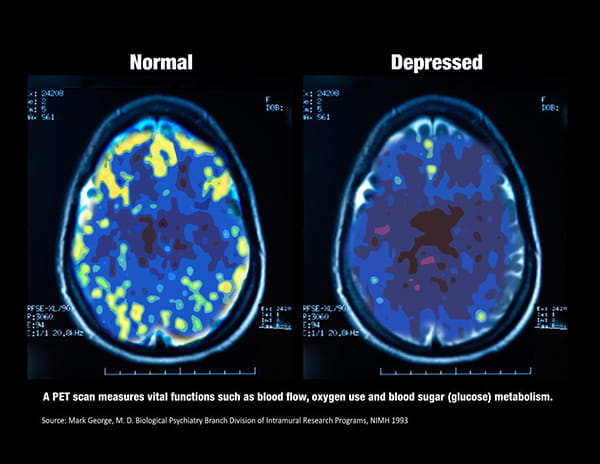
Image: PET scan of non-depressed brain vs. depressed brain. The non-depressed brain is stimulated while the depressed brain has low activity.
You'll be awake throughout the TMS treatment and you can resume daily activities immediately after each treatment. This is not the same as electroconvulsive therapy (ECT). Since there are no effects on alertness or memory, you can drive yourself to and from the appointment.
During the procedure, you will feel the curved device that houses the magnetic coil, resting against your head. You'll also hear a clicking sound that usually lasts for a few seconds.
TMS treatments:
As with any therapy, every person is different and TMS is not for everyone. The most common side effect is mild to moderate scalp pain or discomfort, which usually goes away after the first week of treatment. A TMS-certified psychiatrist will discuss the risks and benefits of treatment with you to determine if you are a candidate.
Transcranial magnetic stimulation therapy is also covered by most insurance providers. Check with your provider about coverage.
1 Dunner DL, et al. J Clin Psychiatry. 2014;75(12):1394-1401
2 ibid
Discover how Main Line Health helps those who struggle with mental illness and substance use live healthy and fulfilling lives.
The neurology team at Main Line Health treats and manages conditions such as migraines, multiple sclerosis, Parkinson’s disease and more.
Discover how Main Line Health neurosurgeons provide expert care, from routine to complex spinal and nervous system conditions for patients.